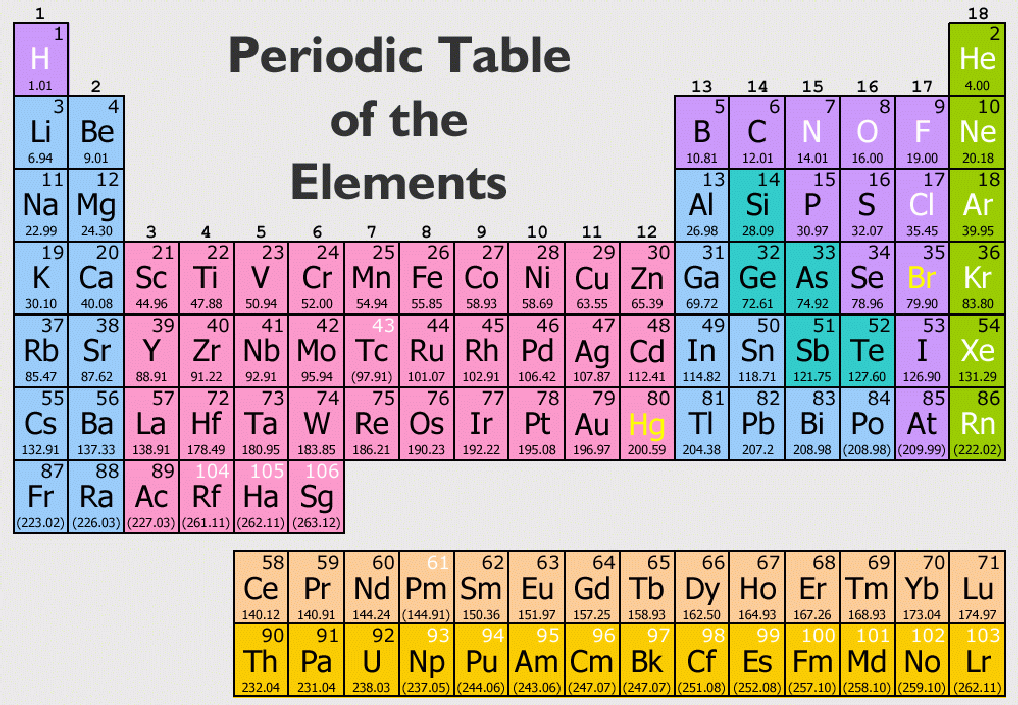
Elements fall somewhere between the least metallic fluorine and the most metallic cesium (or francium, if you include lab-made elements). You can tell how metallic any element is just by finding it on the periodic table. (b) The metallic atomic radius, rmet, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as. Key groups include alkali metals, alkaline earth metals, halogens. The table is divided into metals, nonmetals, and metalloids, each with distinct properties. 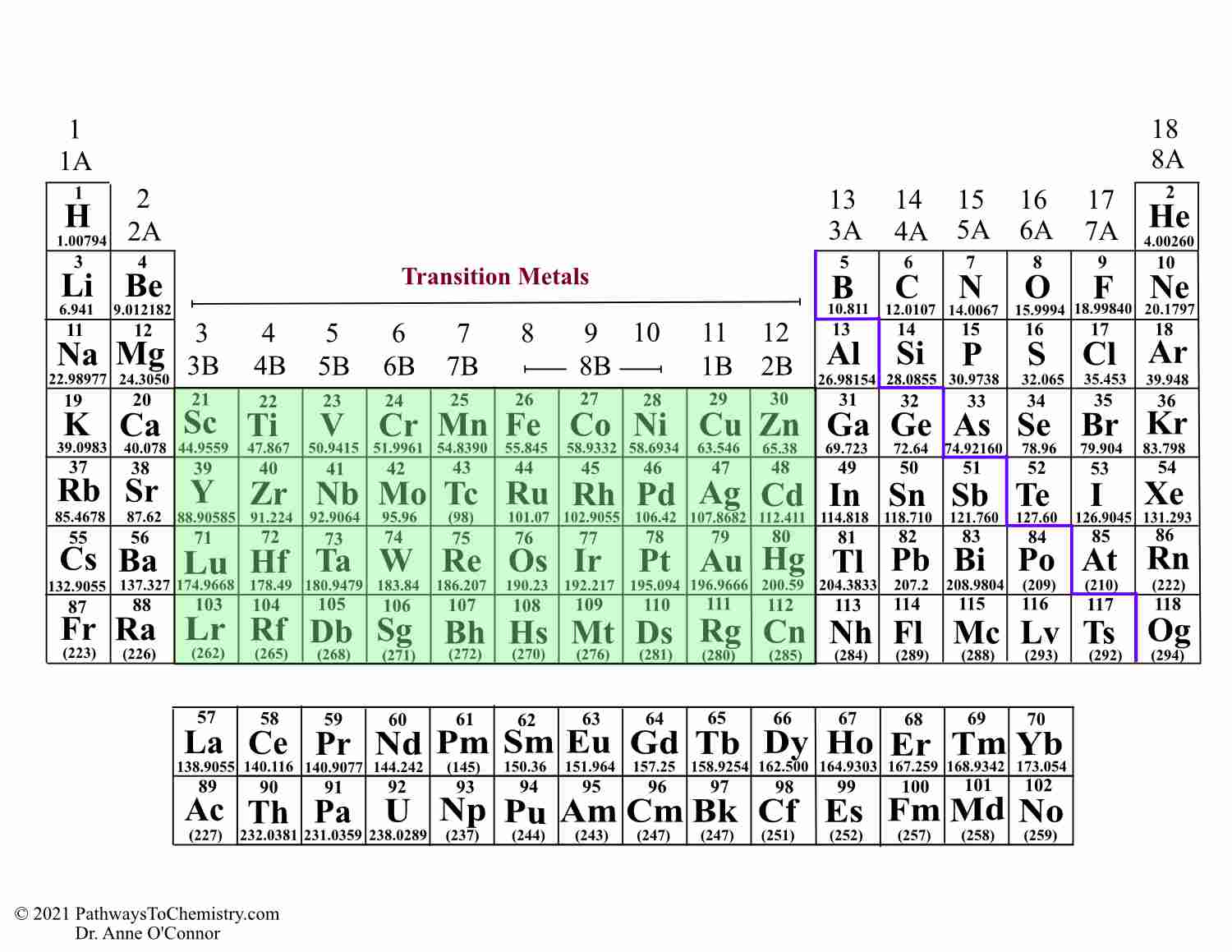
Elements in the same group share similar characteristics, like reactivity. As expected, semimetals exhibit properties intermediate between metals and nonmetals. The periodic table separates metals from nonmetals. (a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. The periodic table organizes elements into groups and periods based on their chemical and physical properties. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Map: Chemistry - The Central Science (Brown et al.) 7: Periodic Properties of the Elements. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The periodic table separates metals from nonmetals. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Semimetals exhibit properties intermediate between those of metals and nonmetals. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. The elements can be broadly divided into metals, nonmetals, and semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
